Chemical Analysis of Solids and Powders
 The Thermo K-Alpha XPS uses a focused beam of x-rays to eject electrons from the near-surface region of a sample in a vacuum.
The Thermo K-Alpha XPS uses a focused beam of x-rays to eject electrons from the near-surface region of a sample in a vacuum.
The energies of those electrons are characteristic of the elements from which they came and the specific chemical bonds that involve those elements.
By plotting the number of photoelectrons as a function of energy, one can obtain detailed information on the near-surface elemental and chemical composition of a sample.
To reserve time or request training on this tool, click here or on the image at left to go to the K-alpha XPS tool page on SUMS.
 |
 |
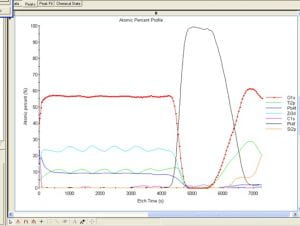 |
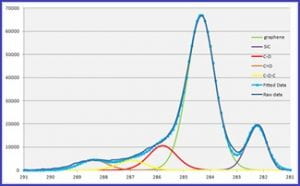
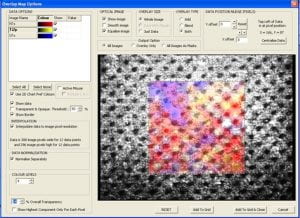
| Deconvolution of Carbon spectrum in a graphene oxide film on SiC shows XPS ability to distinguish chemical states. | Nitrogen-rich areas -in Red on map above – show amine-terminated spots on a Ti layer. The map is overlaid on an optical image of the sample. | An integrated Ar sputter gun allows in-situ sample cleaning and depth profiles like the one above on a multi-layer PZT film. |
Several analysis techniques provide information about elemental composition (XRF, EDS, SIMS, etc.). For inorganic compounds that information is usually enough to determine a sample’s chemical makeup. However, for organic compounds, there are typically numerous chemical permutations even within the constraints of a given composition.
Only a few techniques provide direct evidence of the chemical state of an element. Among them, XPS is unique in it’s surface sensitivity, which makes it an ideal way to study – among other materials – (catalysts, ion-exchange membranes, graphene and other low-dimensional structures, functionalized CNT’s, metal corrosion, etc.)
Sources: Capabilities: Files: Videos: Avantage Data Processing – part 1 Avantage Data Processing – part 2System Details:
-X-ray source: Monochromated Al Kα radiation; Microfocused to a spot with diameter selectable between 30 and 400 μm
-Flood gun: Integrated mixed low-energy electron and Ar-ion gun for charge compensation
-Sputter gun: Ar+ ion gun; Ion energy selectable from 200 to 3000 eV.
-Analyzer: Hemispherical analyzer with MCP detector; Pass energy range from 20 to 250 eV for high- resolution and wide range survey spectra, respectively
Single-point spectra; Line profiles; Area Chemical Maps; Depth Profiles
Samples: Special vacuum-sealed stage for air-sensitive samples; interchangeable plates for flat and powder samples
Thermo K -Alpha Instructions
K-Alpha Depth Profile Instructions
K-Alpha Chemical Mapping Instructions
K-Alpha XPS Sputter rate calculatorPerformance Check Results:
